Get in touch with our team to discuss your requirements
Ecat foundation
- Home
- Ecat foundation
ECAT Foundation – External Quality Assurance You Can Trust

Advancing Laboratory Excellence Through Reliable EQA Programs
At Scorgemi Scientific Solutions, we believe that high-quality diagnostics begin with robust External Quality Assessment (EQA). That is why we proudly support laboratories participating in the ECAT Foundation – a global leader in coagulation EQA schemes, trusted by thousands of clinical laboratories worldwide.
The ECAT Foundation provides structured, evidence-driven assessment programs that help labs evaluate the accuracy, consistency, and reliability of their hemostasis testing.
For laboratories focused on standardization, compliance, and continuous quality improvement, ECAT remains a cornerstone of excellence.
⭐ Why ECAT Foundation Matters for Your Laboratory
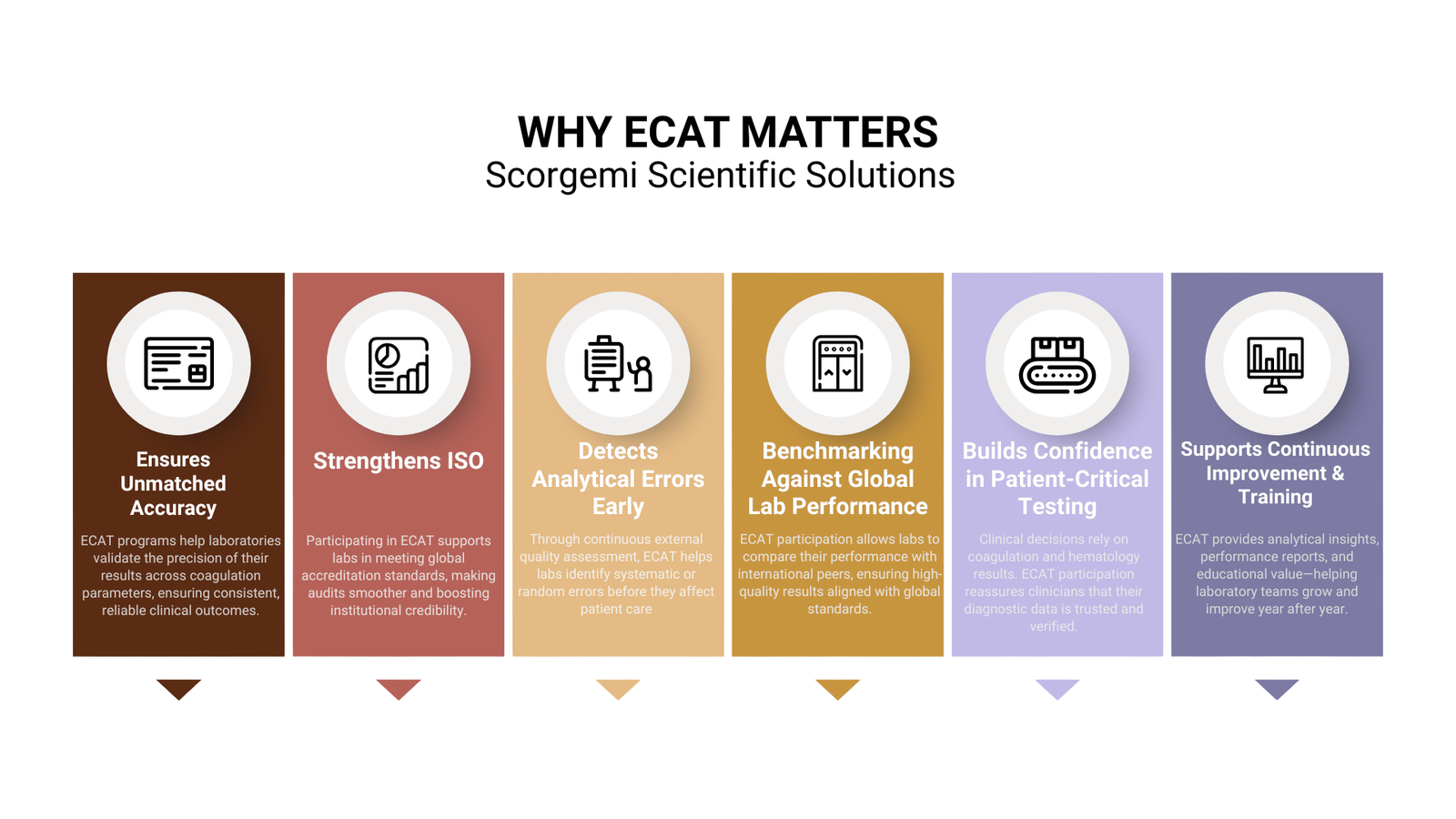
1. Internationally Recognized EQA Programs
ECAT’s quality schemes help laboratories benchmark performance using internationally accepted protocols. This supports accreditation under:
- ISO 15189
- ISO 17043
- NABL standards
2. Precision in Coagulation Diagnostics
The ECAT Foundation specializes in ensuring consistent performance in:
- PT/INR
- APTT
- Fibrinogen
- D-Dimer
- Lupus Anticoagulant
- Factor Assays
- Anti-Xa Heparin
- DOAC Testing
- Thrombin Generation Tests
This aligns seamlessly with Scorgemi’s wide range of reagents, calibrators, controls, and coagulation-specific kits.
3. Strengthens Laboratory Competence
Regular ECAT participation helps labs:
- Reduce diagnostic errors
- Ensure regulatory compliance
- Improve analytical performance
- Build confidence with clinicians and patients
How Scorgemi Supports ECAT Laboratories
Scorgemi Scientific Solutions offers an extensive portfolio of ISO-aligned reagents, calibrators, controls, and analyzers widely used by labs engaged in ECAT programs.
Our product strengths include:
✔ Coagulation reagents & chromogenic substrates
✔ Plasma controls (Normal, Abnormal, LA, L1, L2 etc.)
✔ Calibrators for specialized assays
✔ Thrombosis & hemostasis diagnostic kits
✔ Coagulation analyzers & POCT devices
✔ High-sensitivity substrates for research and routine diagnostics
Our quality systems are aligned with:
- ISO 13485 (Medical Devices – QMS)
- ISO 9001 (Quality Management Systems)
- Traceable manufacturing processes
- End-to-End product validation
This ensures compatibility, reliability, and consistent performance that labs expect when taking part in ECAT EQA cycles.




